Chapter 2 - Classifying Matter
Need access to you textbook? Click here and follow the directions. |
Chapter 2 Key Concepts
Section 1
Section 2
Section 3
Section 1
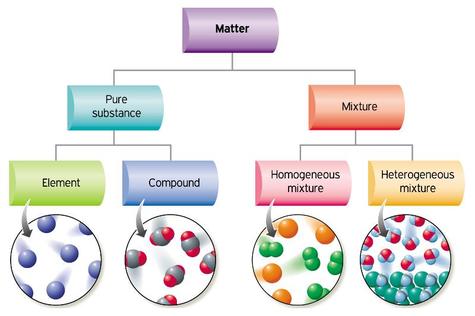
- Why are elements and compounds classified as pure substances?
- How do mixtures differ from pure substances?
- What is the main difference among solutions, suspensions, and colloids?
Section 2
Section 3
Chapter 2 Notes
| Chapter 2 PowerPoint | |
| File Size: | 200 kb |
| File Type: | ppt |
| Chapter 2 Notes Sheet | |
| File Size: | 28 kb |
| File Type: | docx |
Chapter 2 Homework
Section 1
| Section 2.1 Guided Reading | |
| File Size: | 139 kb |
| File Type: | |
Section 2
| Section 2.2 Guided Reading | |
| File Size: | 159 kb |
| File Type: | |
Section 3
| Section 2.3 Guided Reading | |
| File Size: | 148 kb |
| File Type: | |
Chapter 2 Vocabulary
pure substance
element
atom
compound
heterogeneous mixture
homogeneous mixture
solution
suspension
colloid
physical property
viscosity
conductivity
malleability
melting point
boiling point
filtration
distillation
physical change
chemical property
flammability
reactivity
chemical change
precipitate
Chapter 2 Vocabulary on Quizlet - CLICK HERE!
Chapter 2 Journal Activities
| Densities of Liquids Lab | |
| File Size: | 125 kb |
| File Type: | |
| Math Practice - Melting and Boiling Points | |
| File Size: | 143 kb |
| File Type: | |
| Physical and Chemical Change Lab | |
| File Size: | 171 kb |
| File Type: | |
| Forensics Lab | |
| File Size: | 19 kb |
| File Type: | docx |